The purity of polyimide (PI) monomers (primarily dianhydrides and diamines) directly determines the molecular weight, molecular weight distribution, color, thermal stability, and electrical properties of the resulting PI. Electronic-grade and optical-grade PI typically require monomer purity ≥99.9% and metal ions ≤1 ppm. This article systematically introduces mainstream purification methods, applicable scenarios, operational key points, and their respective advantages and disadvantages.
Utilizes the surface adsorption properties of adsorbents such as activated carbon, alumina, silica gel, and molecular sieves to remove colored impurities, polar impurities, and trace metal ions. Common adsorbents and their applications are shown in the table below:

Adsorption methods offer efficient decolorization and metal ion removal under mild conditions at low cost. However, they require large adsorbent quantities, present recovery challenges, and incur some product loss.
Utilizes ion exchange resins (cationic, anionic, chelating types) to exchange with metal ions (Na+, K+, Fe3+, Cu2+, etc.) in monomer solutions, achieving deep removal. Suitable for high-end applications requiring metal ions ≤1ppm, such as electronic-grade and optical-grade PI monomers. Any requirement for low metal ions, low chloride ions, and low acidity/alkalinity necessitates ion exchange. Often combined with recrystallization and adsorption methods, it avoids damaging monomers or introducing new impurities, serving as the essential final purification step for high-end PI raw materials. The process is relatively straightforward: pass the monomer solution through ion exchange columns + chelation resin columns while controlling flow rate, temperature, and pH to ensure complete metal ion exchange. Subsequently, elute and regenerate the resins for reuse. Common resin types for PI monomers are as follows:
Cation Exchange Resin (H+ Type): Removes all metal cations, the most common and critical type, suitable for both dianhydrides and diamines.
Anion Exchange Resin (OH- Type): Removes Cl-, Br-, SO42-, and organic acid anions, used for trace acids and halide impurities in dianhydrides.
Chelating Resin: Exhibits extremely high selectivity for transition metals (Fe, Cu, Ni), dedicated for electronic-grade/ultra-low metal ion content monomers.
Industrial practice commonly employs a series combination of cation resin + chelating resin + anion resin. The ion exchange resin method achieves exceptionally high metal ion removal rates (down to ppb levels), excellent selectivity, and continuous operation. However, resin costs are high, regeneration is required, processing capacity is limited, and solvents must be anhydrous.

Chromatography is suitable for preparing high-purity reference standards (purity > 99.99%) in laboratories or separating structurally very similar isomers (e.g., certain dianhydrides or diamine monomers with positional isomers). This method offers extremely high separation efficiency and purity limits, but compared to the previous methods, it has low throughput, extremely high costs, and is unsuitable for industrial-scale production. Chromatography encompasses three main techniques: column chromatography, high-performance liquid chromatography (HPLC), and gas chromatography (GC). Column chromatography uses silica gel, alumina, or ion-exchange resins as stationary phases; HPLC employs reverse-phase C18 or normal-phase silica gel for analytical and small-scale preparative applications; while GC is suitable for analyzing and micro-purifying volatile, thermally stable monomers.
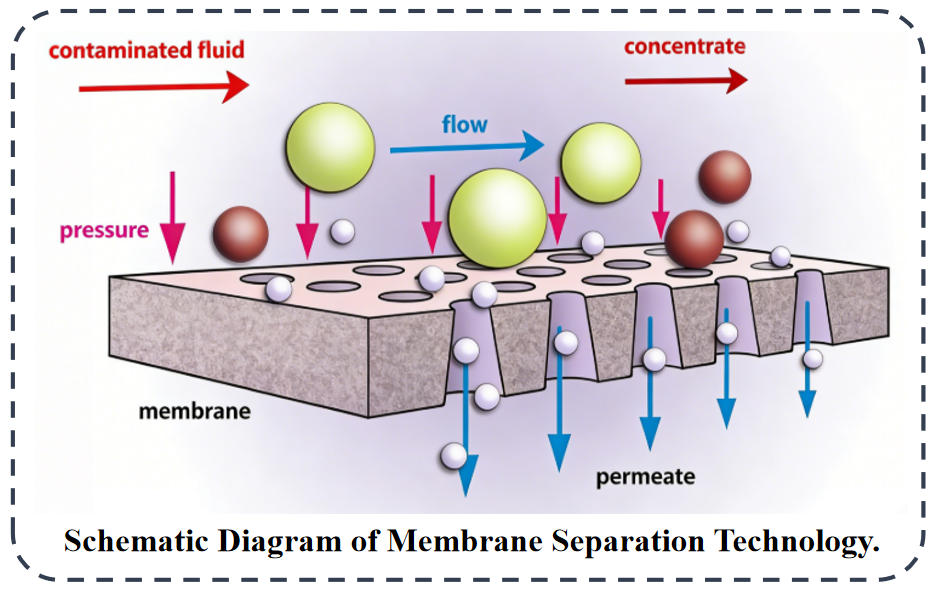
Membrane separation achieves selective separation by leveraging differences in “sieving + dissolution diffusion” across membranes under pressure, based on variations in molecular weight, polarity, charge, and particle size. This method operates without high temperatures, phase changes, or significant energy consumption, preserving monomers and being suitable for heat-sensitive substances. It allows for continuous, automated processes and can be combined with recrystallization or melt crystallization to substantially enhance purity and yield. However, membranes are costly, prone to fouling, and demand high solvent compatibility. Membrane materials applicable for PI monomer purification include:
Ultrafiltration (UF) Membranes: Retain macromolecules, oligomers, colloids, pigments, and insoluble impurities; permeate monomers, salts, and small molecules. Used for crude purification, pretreatment, turbidity removal, and decolorization.
Nanofiltration (NF) Membranes: Retain polyvalent ions, certain metal ions, oligomers, and colored impurities; permeate small-molecule monomers. Suitable for desalting, partial metal ion removal, and pre-purification.
Reverse Osmosis (RO) Membranes: Retain nearly all salts, metal ions, and small-molecule impurities; primarily permeate solvents. Used for solvent recovery, concentration, and deep desalting.
Organic Solvent Nanofiltration (OSN): Specifically designed for non-aqueous systems, serving as the core membrane technology for PI monomer purification. Resistant to organic solvents like DMF, NMP, DMAc, and methanol. Retains oligomers, pigments, metal complexes, and macromolecular impurities; permeates dianhydrides and diamine monomers. Used for refining electronic-grade PI monomers, removing oligomers, decolorization, and metal ion removal.
PI monomer purification centers on recrystallization, supplemented by adsorption, ion exchange, distillation/melt crystallization, and other methods to achieve purity upgrades from industrial to electronic grade. In actual production, 2~3 methods are combined based on monomer structure, impurity types, purity requirements, and cost. For general industrial-grade PI, extraction + activated carbon adsorption + recrystallization is commonly used; for electronic/optical-grade PI, activated carbon or molecular sieve adsorption + ion exchange resin + recrystallization is preferred. Strict control of solvents, environment, and equipment is essential to consistently obtain the high-purity monomers required for high-performance finished PI products.
Contact us to learn more about our advanced electronic chemicals and speciality polymer materials, and how they can enhance your production performances.