The purity of polyimide (PI) monomers (primarily dianhydrides and diamines) directly determines the molecular weight, molecular weight distribution, color, thermal stability, and electrical properties of the resulting PI. Electronic-grade and optical-grade PI typically require monomer purity ≥99.9% and metal ions ≤1 ppm. This article systematically introduces mainstream purification methods, their applicable scenarios, operational key points, and respective advantages and disadvantages.

The recrystallization method utilizes the difference in solubility of monomers and impurities in solvents with temperature. It achieves solid-liquid separation and purification through the following steps: heating to dissolve → hot filtration to remove insoluble impurities → cooling and adding counter-solvent to precipitate monomer crystals. This method is suitable for dianhydride monomers (PMDA, BPDA, ODPA, 6FDA) and diamine monomers (ODA, PDA, BAPP, TFMB, BAPB), as well as impurities such as unreacted starting materials, isomers, oligomers, inorganic salts, and colored impurities. For diamines, a typical recrystallization workflow is as follows:
First, dissolve the crude product in a solvent (e.g., ethanol, isopropanol, acetonitrile, toluene) and reflux until complete dissolution. Add activated carbon or activated alumina, reflux for 0.5~2 hours to perform decolorization and adsorption, then hot-filter to remove the adsorbent and insolubles. Slowly cool or add a counter-solvent (water, hexane) to perform isothermal recrystallization (5~10oC, 2~12 h), promoting the growth of large, high-purity crystals. Finally, perform vacuum filtration or centrifuge, wash the crystals 2~3 times with cold solvent, and vacuum dry (80~150oC, -0.09 MPa) to remove residual solvent.
Solvent selection is critical in this method. Solvent systems for selected monomers are shown in the table below:

Recrystallization offers simple operation, low cost, high yield, and scalability; repeated recrystallization can enhance purity. However, this method has limited effectiveness for separating isomers with highly similar structures, requires strict control of solvent residues, and involves relatively high energy consumption.
This method utilizes differences in distribution coefficients between monomers and impurities in solvents of varying polarity. Impurities are removed via liquid-liquid extraction or solid-liquid washing. It is suitable for pre-treatment of crude products to remove large amounts of unreacted raw materials, inorganic salts, and water-soluble impurities. When combined with recrystallization, it can significantly reduce recrystallization load. Common approaches include the following three:
Liquid-Liquid Extraction: The monomer is dissolved in an organic solvent and washed with an acid/base/salt solution to remove amine or acid impurities and metal ions.
Soxhlet Extraction: The solid crude product undergoes continuous reflux extraction with a solvent to remove low-boiling-point impurities and tar.
Pulp Washing: The crude monomer is mixed and stirred with an unsuitable solvent, then filtered to remove surface-adsorbed impurities.
Solvent extraction/washing offers mild operation, easily controllable conditions, and suitability for large-scale crude purification. However, its purity improvement is limited and requires combination with recrystallization or distillation.
This method utilizes boiling point differences between monomers and impurities for separation via vacuum/atmospheric distillation; Suitable for monomers with boiling points ≤350oC. Examples include p-benzenediamine, hexamethylenediamine, isophthalamide, terephthalic acid dimaleimide (PMDA), and certain aliphatic diamines. The industrial process for purifying hexamethylenediamine via distillation is now well-established: First, crude product undergoes dehydration and decoking. Subsequently, a continuous distillation column removes light fractions → removes heavy fractions → performs product distillation (vacuum range 1~10 mmHg, temperature 180~250oC). The resulting ethylenediamine achieves 99.9%~99.99% purity with a 90%~95% yield.
Distillation/vacuum distillation offers high process continuity, superior product purity, and suitability for large-scale production, effectively removing metal ions and inorganic salts. However, it requires significant capital investment, high energy consumption, and strict temperature control in an oxygen-free environment to prevent partial dianhydride hydrolysis or diamine oxidation at elevated temperatures.
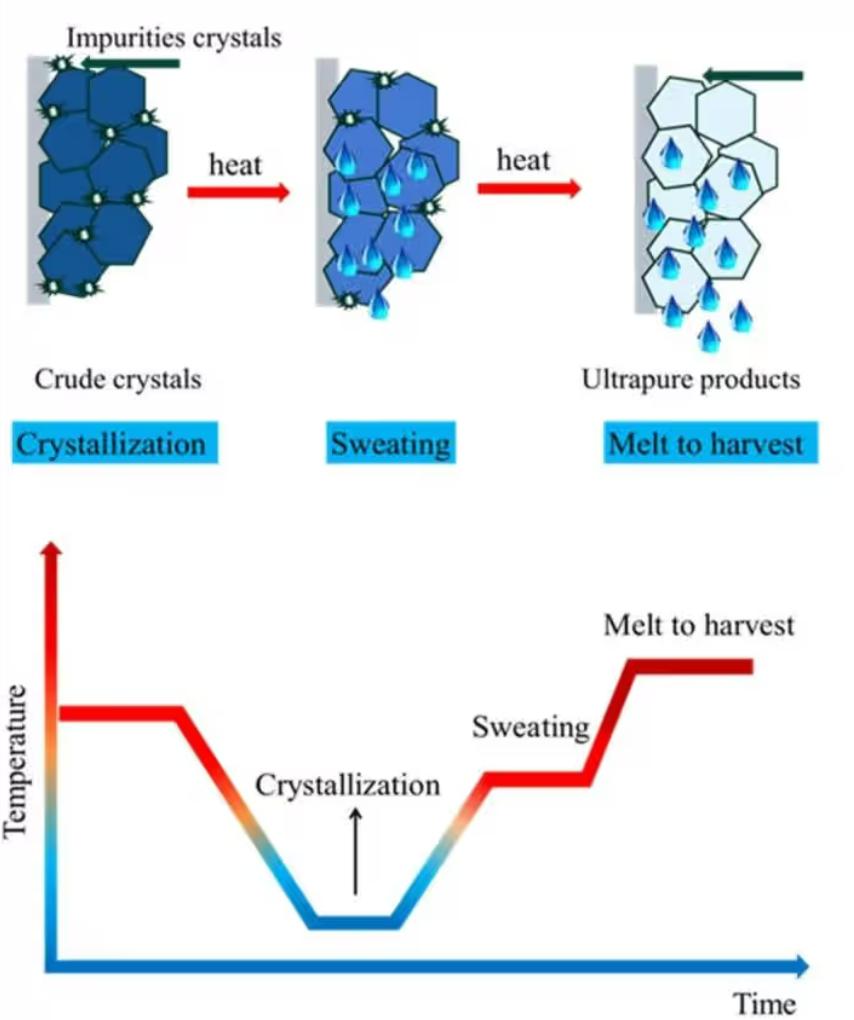
Melting crystallization exploits the melting point differences between monomers and impurities. Through stepwise cooling or melting, high-purity crystals precipitate preferentially while impurities accumulate in the mother liquor. This method is suitable for thermally stable monomers with melting points between 50~200oC, such as hexamethylene diamine and certain dianhydride monomers. The complete industrial process (standard three-step method) is now well-established:
Melting: Crude monomer is introduced into the crystallizer and heated to 3~10oC above its melting point until fully molten. Stirring ensures uniformity and degassing. For diamines, N2 protection is recommended to prevent oxidation and yellowing.
Crystallization: Cool extremely slowly (0.1~2oC/h; slower cooling yields more regular crystals and higher purity). A dense crystalline layer forms first on the tube walls or plate surfaces. High-purity components preferentially enter the crystal lattice, while low-melting-point, structurally incompatible impurities remain in the melt.
Sweating (Critical Step): Slightly raise the temperature to expel impurities from the crystals. Maintain this slightly elevated temperature (1~5oC below the melting point) for 0.5~3 hours. Impurities at grain boundaries and defects melt and drip out first, making the crystal interior denser. Purity increases significantly, and the melting range narrows markedly.
Finally, the impurity-laden mother liquor is drained off. The pure crystals are then remelted, drained, cooled, and crushed to yield high-purity PI monomer. The final product achieves 99.99% purity with a 90~95% yield.
This method replaces distillation, substantially reducing energy consumption while offering solvent-free operation, high purity, and suitability for heat-sensitive monomers. However, it requires complex equipment, involves a lengthy cycle, and has limited effectiveness for separating impurities with similar melting points
Contact us to learn more about our advanced electronic chemicals and speciality polymer materials, and how they can enhance your production performances.